He saw the tests as a violation of 'trusting, desperate human beings'
 = PDF file size may be large. Document titles are listed at the bottom of the page. (Due to the poor quality of some of the original documents, some PDFs might be difficult to read. Highlighting was done by reporters.)
= PDF file size may be large. Document titles are listed at the bottom of the page. (Due to the poor quality of some of the original documents, some PDFs might be difficult to read. Highlighting was done by reporters.) |
| Refer to Using this Site for more information. |
|
By Duff Wilson and David Heath
Seattle Times staff reporters
Copyright © 2001 The Seattle Times Company
FOR NEARLY two decades, Dr. John Pesando sounded the alarm over what he saw as a dangerous and unethical human experiment at Seattle's Fred Hutchinson Cancer Research Center.
In the 1980s, he complained from the inside: As a member of an oversight committee at "The Hutch," he repeatedly objected to the clinical trial in which leukemia patients were dying at unusually high rates.
In the 1990s, he complained from the outside: In letter after letter - to members of Congress, to federal and state agencies, to newspapers - he said "many good people needlessly lost their lives" in the leukemia experiment, which lasted from 1981 to 1993.
"Trusting, desperate human beings were used as laboratory rats," he wrote to state authorities in 1994. "One should not assume that these patients would have soon died anyway because they had cancer, and that it was therefore not possible to do any harm. Many of these patients were correctly told that they could expect cure rates of approximately 50 percent (with standard treatment) ...
"Real people lost their lives, and there was no way to stop it."
 What happened
What happened
A timeline of doctors' complaints and officials' responses. |
|
For the most part, Pesando was received as Hutchinson Center officials have portrayed him: a disgruntled former employee with an ax to grind. But a Seattle Times investigation supports most of his allegations and finds his concerns shared by some of the most respected cancer doctors in the Northwest.
Seventeen years passed between the time Pesando and other doctors first objected to Protocol 126, as the experiment was labeled, and yesterday's revelations in The Times of deaths, misleading assurances and potential financial conflicts surrounding the experiment.
In those years, the doctors' complaints were largely rebuffed, rejected or ignored. Finally, under incessant pressure from Pesando, they were taken up by federal and state agencies but never fully investigated.
Underlying all of this, Pesando and other doctors believe, was a reluctance to challenge an institution as respected and powerful as the Hutchinson Center. That reluctance was all the greater because one of the people behind Protocol 126 was Dr. E. Donnall Thomas, a Hutch co-founder and co-winner of the 1990 Nobel Prize in Medicine.
As Pesando puts it: "They were all afraid of taking on the 800-pound gorilla."
Experiment already under way
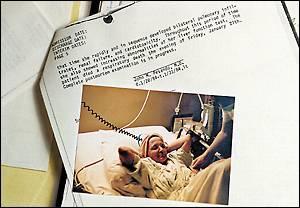 |
| Harley Soltes / The Seattle Times |
| A snapshot of Ruth Fisher who died in January 1984 after a graft failure and second transplant |
John Pesando came to Seattle and The Hutch in 1982 as a 36-year-old magna cum laude graduate of Harvard University with a medical degree and Ph.D. from the Albert Einstein College of Medicine in New York. Pesando was board-certified in oncology and internal medicine; his research focused on tumor immunology. He had taught at Harvard and had worked for four years as a cancer doctor and researcher at the Dana Farber Medical Center in Boston.
At Harvard, Pesando had discovered an important antibody marker in tracking and treating leukemia, and conducted the first-ever clinical trial with a monoclonal antibody, treating five leukemia patients.
He was recruited to The Hutch by one of its co-founders, Dr. Rainer Storb. Soon after joining the staff, Pesando was asked by his bosses to serve on the Center's Institutional Review Board.
That board was required by federal law to review all proposed human experiments at The Hutch. Its members were to ensure that risks to patients were minimized in relation to potential benefits, and that patients fully understood those risks before agreeing to participate in clinical trials.
Pesando was reluctant to take on the unpaid duty of policing his colleagues, some of whom were in a position to affect his future. But he believed deeply in the need for such policing, he says, so he swallowed hard and served.
Also joining the review board was a new chairman, Dr. Henry Kaplan, from Seattle's Swedish Medical Center, where some Hutch patients were treated. He is now one of the Northwest's most highly regarded oncologists.
From their first days on the board together, Pesando and Kaplan were concerned about Hutch researchers using unproven new antibody drugs.
In Protocol 126, researchers were testing antibodies to see if the elimination of certain cells in the blood, called "T-cells," would make bone-marrow transplants safer. The researchers theorized that killing off the T-cells in the marrow of donors before injecting it into patients would help stave off a troublesome complication known as graft-versus-host disease, or GVHD.
The experiment was directed by Don Thomas; Dr. John Hansen, head of a tissue-matching lab and later clinical director; and Dr. Paul Martin, a young oncologist. The trial was funded by the National Cancer Institute.
From the time the experiment was first brought to the oversight committee early in 1981, some committee members had challenged it, saying:
•The science behind the experiment was dubious. While mice benefited, some felt there had been inadequate prior research on dogs before moving on to tests in humans.
•Some thought T-cell removal might actually prevent donated bone marrow from engrafting, or taking hold in the recipient's bones, and some thought it might open the way for the return of the cancer.
•While GVHD was bothersome and uncomfortable, it was seldom fatal. And the proposed subjects of Protocol 126 - those with siblings whose tissue type matched theirs - were the least likely to get it. Besides, there were other ways to effectively treat GVHD.
•Unlike most cutting-edge medical research, Protocol 126 proposed experimenting on the healthiest, rather than the sickest, patients. Committee members saw no need to subject those patients to undue risk.
•The "informed-consent" form for the experiment - the required document outlining risks for patients - was misleading.
Despite all these concerns, the previous oversight board had approved the experiment in April 1981. By the time Pesando and Kaplan joined the board, the experiment was well under way.
Financial conflicts of interest
Within their first few months on the Institutional Review Board (IRB), the new members suspected that Protocol 126 wasn't working. Patients enrolled in the experiment were dying - including some people who otherwise would be expected to live.
Kaplan and Pesando raised many of the same questions their predecessors had on the medical and scientific aspects of the experiment. They received lots of assurances but no answers.
 Meanwhile, a new concern emerged: Rumors were rampant in hospital hallways that the researchers - Thomas, Hansen and Martin - all had financial interests in Genetic Systems, the Seattle biotechnology company that owned rights to some of the drugs being tested in the experiment. Meanwhile, a new concern emerged: Rumors were rampant in hospital hallways that the researchers - Thomas, Hansen and Martin - all had financial interests in Genetic Systems, the Seattle biotechnology company that owned rights to some of the drugs being tested in the experiment.
When Kaplan asked Hutch officials about the rumors, he was told the researchers had no financial conflicts. What he was not told was that not only did those researchers all hold stock in Genetic Systems, they all held advisory or actual working positions with the company. Nor was he told that The Hutch itself had stock in Genetic Systems.
"We didn't even know which investigators were connected with which companies," Kaplan recalls now. "We never got the opportunity to ask about the decision-making process. I guess the fair thing to say was John (Pesando) was absolutely convinced there was a problem, and the rest of us thought there was enough to raise questions about. They were not answered satisfactorily in the time I was on the board."
Meanwhile, the early stages of Protocol 126 were proving a failure, and patients were dying - some, apparently, as a direct result of the experiment. Among the dead were two patients Pesando himself had treated.
When their two-year terms on the Institutional Review Board ended in 1985, Kaplan and Pesando were told again by the president of The Hutch, Dr. Robert Day, that their concerns about both medical and ethical problems with Protocol 126 would soon be addressed.
They trusted that assurance, and went on to focus on their careers. Kaplan built one of the most successful private oncology practices in the Northwest; last year, he was the cover subject of a Seattle Magazine feature entitled "Top Doctors."
Career hits a snag
Pesando, meanwhile, went about trying to land a long-term position doing cancer research at The Hutch. That didn't happen. In 1987, his contract expired and was not renewed. Storb says Pesando "didn't work out. He was removed by Don (Thomas) for a number of reasons. I agreed with them. He was not an active member. He led a sheltered life. He was very paranoid about sharing data. He didn't bring in grants. He left under poor circumstances."
Pesando has another explanation: his run-ins with Thomas and the other people behind Protocol 126. He recalls Thomas confronting him after one particularly tense session, barking: "Who the hell are YOU to question what we do here?"
(Thomas refused to be interviewed by The Times for the articles running this week.)
Pesando doesn't deny that he struggled at The Hutch. He says that was because he was unable to get support from his bosses for research he proposed.
He tried unsuccessfully for a time to find a good position elsewhere in his specialty areas of leukemia and lymphoma - "They (would) go to The Hutch for recommendations" - then decided to take another path. He began working as a consultant for the Social Security Administration and for biotechnology companies.
He and his wife settled into a hundred-year-old Victorian home on Capitol Hill and raised their daughter.
Over the years, Pesando tried to put Protocol 126 out of his mind. He might have succeeded, had he not picked up a book entitled "Magic Bullets" in 1991. In it, Seattle author Grant Fjermedal describes the invention of new antibodies to seek out and destroy cancer.
Among the companies Fjermedal wrote about was Genetic Systems. He outlined the company's relationship with The Hutch and its doctors, and quoted Day, Hutch president, on why such financial interplay was a positive factor in medical research, not a negative.
Pesando was incensed to see this apparent confirmation of what he and Kaplan had suspected all along: that Protocol 126 was driven not only by science but also by money. And Day's position was a signal that nothing had changed.
Pesando contacted Fjermedal, a longtime science and medical writer, who was struck by Pesando's knowledge of the situation and by his passion.
"What you've got in this guy is a rarity," Fjermedal said in a recent interview. "You've got somebody who has the credentials. He's seen all this, he's on the inside, and he's coming forward. That's very, very rare."
Fjermedal encouraged Pesando to take his complaints to the proper authorities.
For the rest of the decade, that's precisely what this lonely warrior did - only to be repeatedly frustrated.
Federal agencies take a look

President Day successfully defended The Hutch, but in the end a state official said, 'There still remained some doubt in our mind.' |
Pesando began firing off letters in various directions in 1991. He wrote to members of Congress, to federal and state agencies, even to The New York Times.
Finally, a letter to the federal Office of Protection from Research Risks (OPRR), a unit of the Department of Health and Human Services, caught someone's attention.
"Basically," Pesando wrote, "senior clinicians at the Fred Hutchinson Cancer Research Center in Seattle conducted clinical trials with high therapy-induced mortality rates while they were major stockholders in the company with commercialization rights to those therapies."
Admitting to feeling "much anger," Pesando concluded, "The seriousness of this issue and its implications for future patient care continue to make it very difficult for me to take the expedient course of doing nothing."
In May 1993, the OPRR opened an investigation of the case. Thomas Puglisi, chief of the agency's compliance department, assigned investigator Kamal Mittal.
Mittal got Pesando's permission to use his name, and three months later, sent a copy of Pesando's letter to Day, the Hutchinson Center president.
Two months after that, Day responded with a 24-page letter and 2-inch stack of attachments. In that package, he defended The Hutch and its researchers vigorously.
He insisted that a successful outcome to Protocol 126 would not have benefited Genetic Systems or its stockholders - ignoring the fact that some of the chemicals being tested were licensed to Genetic Systems, and that the very existence of the experiment had increased the company's value.
He insisted that any risks "were fully disclosed to and discussed with the patients." However, the consent form underplayed the risk of graft failure and failed to mention the likely outcome - death - if a second transplant were necessary.
He insisted that the IRB had the power to address any concerns - ignoring the fact that Pesando and Kaplan had pushed for an outside review.
And he insisted that concerns about conflicts of interest "were addressed promptly" by a policy adopted by the Hutch Board of Trustees in March 1983. That policy barred employees from participating in research in which they or a family member had an economic interest of any type. However, the policy was not enforced and resulted in no changes in Protocol 126. In fact, Hansen and Martin say they were never told about it.
(Day, who retired as Hutch president in 1997, refused to be interviewed by The Times for the articles running this week.)
The Hutch also hired the best medical-ethics defense lawyer at the then-biggest law firm in Washington, D.C.: Barbara Mishkin of Hogan & Hartson, who phoned Puglisi and met with Mittal briefly.
Pesando had no opportunity to respond to Day's claims. He called the OPRR several times to offer help and information but never heard back.
Frustrated, he went to the top, writing to Health and Human Services Secretary Donna Shalala. The result: more frustration.
Pesando did receive a response, from Wendy Baldwin, acting deputy director for extramural research at the Public Health Service. Baldwin wrote that she recognized "the very serious nature of the issues you have raised" and promised to review the case.
But that promise was an empty one, e-mail released to The Times under the Freedom of Information Act shows.
That e-mail, between Puglisi of the OPRR - the actual author of the letter signed by Baldwin - and Dr. George Galasso of the National Institutes of Health, indicates that top federal officials had already decided there was "nothing wrong" with conflict-of-interest issues in Protocol 126. An e-mail from Galasso suggests they send it to another office as "sort of CYA (cover your ass)."
Investigation is dropped
Meanwhile, other federal documents show, the primary investigator, Mittal, was far from convinced there was no problem. He had several pointed questions he wanted to ask Hutch officials:
•Did the IRB feel compromised or strong-armed?
•Was the risk/benefit ratio in favor of the patients?
•Were patients fully and correctly informed of risks?
•Did the doctors violate the conflict-of-interest policies? (Mittal added: "Answer appears to be yes!")
•Was it proper to develop products with federal funds that would profit a private company and doctors?
"To get answers to these questions, it may be necessary to arrange a site visit to speak with various individuals including the members of the IRB at the time period in question," Mittal wrote.
But then Mittal, who'd been brought in as temporary help, asked to be transferred to another job and was removed from the case.
"I left it almost in the middle," Mittal recalls.
Pesando's complaint lay untouched in Washington, D.C., for a year after that.
"It was just a matter of our workload being overwhelming at the time," Puglisi said.
The OPRR was absorbed in a fertility-clinic scandal in California. It was chronically understaffed. At one time it had about 70 open investigations being conducted by one full-time professional staff member and two or three part-timers, the House Committee on Government Reform found.
Eventually, Pesando and Day both wrote, separately, to ask for a progress report. Puglisi assigned a senior staffer, William Dommel, to write a report based on information in the file, nearly all of it provided by The Hutch.
That report, dated Sept. 5, 1995, and signed by Puglisi, concluded that the complaint was "unsubstantiated" and that The Hutch was not at fault since its own Institutional Review Board had failed to stop the activities. The OPRR blamed the Hutch IRB for not doing more to stop the study. The OPRR blamed the Hutch IRB for not doing more to stop the study.
The federal investigators never interviewed Pesando or Kaplan, both of whom had tried to stem the study, or any other IRB members.
They never interviewed Thomas, Hansen, Martin, Day or any other Hutch officials.
They never interviewed any of the families of the trial subjects.
They never asked for a single tape of a single IRB meeting, nor ever spoke with a single outside expert on the risks of T-cell depletion and second transplants.
Pesando was stunned.
He wrote again to HHS Secretary Shalala and to Harold Varmus, director of the National Institutes of Health. Steam rose from his rhetoric.
"In late 20th century America, prominent physicians at a major cancer center knowingly risked the lives of unsuspecting patients in pursuit of financial gain, successfully bypassed regulatory bodies, and repeatedly silenced opposition. ... Yet there could hardly be less concern if laboratory rats had died instead."
He called the investigation "arrogant, chilling and totally unacceptable."
Shalala and Varmus never replied. Baldwin of the Public Health Service wrote to Pesando on their behalf.
"After careful examination of the OPRR's findings, I am convinced that the investigation was exhaustive and conducted with objectivity and due consideration for the rights and welfare of human subjects involved in research," Baldwin wrote.
A fax obtained by The Times, however, suggests there was no such "careful examination" by Baldwin. Instead, it shows that Dommel - the very person who wrote the findings about which Pesando was complaining - had drafted the response for Baldwin to sign. Instead, it shows that Dommel - the very person who wrote the findings about which Pesando was complaining - had drafted the response for Baldwin to sign.
Pesando wrote to Varmus again: "The NIH's handling of this matter suggests that it was afraid of finding something, and the profound apathy of those at the top to misconduct in the field helps to explain why such problems exist."
This time, Pesando got no response.
State panel takes on the case
While he was waiting for Washington, D.C., to act, Pesando took his crusade to Washington state officials.
He wrote to Dr. Larry Bryce, chairman of the state Department of Health's Medical Quality Assurance Commission, in December 1994, repeating his complaint:
"In essence, financial conflict of interest led to highly unethical human experimentation which resulted in at least two dozen patient deaths. Oversight committees were misled, lied to, and kept uninformed while in an atmosphere of fear and intimidation ...
"I have done my part, and more, to correct this problem. I would like to see someone else show some interest."
The state commission showed some interest in the complaint but decided to wait for the federal findings.
Nine months after federal officials dropped the case, the state opened its own investigation. In June 1996, a year and a half after Pesando's filing, the case was assigned to Dr. Robert Miller, physician staff member of the state commission, and investigators James Smith and Bill Crowell.
Their investigation went much further than the feds' had. They interviewed Hutch officials, Kaplan and Pesando. and Pesando.
Kaplan had forgotten many details of his own role by then. For instance, he said he wrote one letter of objection, rather than the three The Times has found, and received just "a one-sentence letter of reply" from Thomas, rather than the lengthy missives Thomas, Day and Dr. Frederick Appelbaum, head of The Hutch's clinical-research division, sent him in the mid-1980s.
The state investigators didn't interview the researchers involved but obtained written statements from Thomas and Hansen.
Thomas' letter, marked "CONFIDENTIAL," was his only written response since the flat denial of conflict in 1984.
"The decision as to whether or not these studies should or should not continue was not made by me," Thomas wrote.
Storb and Pesando say Thomas, though technically not a member of the review panels, was influential in continuing the studies.
"I have never exposed any patient to treatment risks for reasons which were not ethically and medically appropriate, and I did not do so in the matter into which you are inquiring," Thomas wrote.
In his letter, Hansen noted he was not the principal investigator on Protocol 126, and in fact had "no involvement" in it while working for Genetic Systems.
However, Hansen was named as the second author in several papers on the clinical trial. He was Martin's mentor and supervisor. And he received copies of all the key correspondence from The Hutch even while he was working at the private company holding commercial licenses for the antibodies.
In September 1997, with the case still unresolved, Miller left the state Medical Quality Assurance Commission and its investigating team.
With his departure, this investigation died.
The commission closed the complaint, citing "no cause for action," in January 1998, three years after Pesando's letter.
Miller says he was "saddened and alarmed" by the lack of follow through.
At a minimum, he said in an interview, the commission should have acted on ethical violations by Thomas and Hansen for influencing a study of materials licensed to the company in which they owned stock.
"To me, what Dr. Thomas did and the other doctor did was a clear conflict of interest," Miller said in an interview. "I think that's a pretty obvious, well-known ethical principle."
Dr. David Williams, an internist who chaired the state Medical Quality Assurance Commission at the time, was one of two doctors who personally reviewed the case. He says now that he, too, was not satisfied with the outcome.
"There still remained some doubt in our mind," he said. "I was never truly satisfied that the protocol had been written in a manner that was truly appropriate."
'Just trying to do our duty'
Through it all - unresponsive bureaucrats, half-baked investigations, patronizing letters - John Pesando, now age 55, persisted. He was both haunted and motivated, he says, by the memory of two of his patients who died in the experiment - Ruth Fisher, a 40-year-old computer programmer from California, and Jacqueline Couch, a 32-year-old lawyer for the city of New York.
He took it personally when people died on his watch and when he was, in his view, pushed out of The Hutch for raising his voice in favor of ethics and patient safety.
His quixotic quest both frustrated and frightened his wife, Patricia, who worried about his reputation.
But he kept pushing. Pushing until he got the attention of reporters at The Seattle Times, who took up the investigation.
Survivors of patients who died in Protocol 126 are grateful for that persistence.
Pete Wright of Heflin, Ala., whose wife, Becky, died in 1987, said: "I might slough this off to sour grapes if I didn't know Dr. Pesando. But I have nothing but respect for him."
Wright says that while caring for his wife, Pesando never talked about conflicts of interest or research misconduct, though he did try to talk them out of enrolling in the experiment on medical grounds.
"We were not looking for trouble," Pesando said. "We were just trying to do our duty and take care of patients."
Duff Wilson's phone number is 206-464-2288. His e-mail address is dwilson@seattletimes.com.
David Heath's phone message number is 206-464-2136. His e-mail address is dheath@seattletimes.com.
 |
|
Dr. John Pesando's complaint to the Office for Protection from Research Risks, May 14, 1993 (1.1MB PDF)
|
 |
|
Hutch Administrator Dr. Robert Day's response to the complaint, Oct. 18, 1993 (7MB PDF)
|
 |
|
E-mail showing federal officials deciding "nothing wrong" with undisclosed financial interests, Dec. 10, 1993 (56K PDF)
|
 |
|
Federal investigator Kamal Mittal's analysis, questions and recommended course of action, Dec. 8, 1993 (2.2MB PDF)
|
 |
|
OPRR closes the case, Sept. 5, 1995 (1MB PDF)
|
 |
|
Dr. Pesando writes two top federal officials, Dec. 01, 1995 (548K PDF)
|
 |
|
National Cancer Institute aide responds to Pesando, Jan. 11, 1996 (136K PDF)
|
 |
|
Fax shows OPRR wrote that letter for NCI, December 6, 1995 (140K PDF)
|
 |
|
Dr. Pesando writes Dr. Harold Varmus again, Oct. 16, 1996 (264K PDF)
|
 |
|
Dr. Henry Kaplan's letter to state investigator, Sept. 6, 1996 (332K PDF)
|
 |
|
Dr. John Pesando's letter to state investigator, Sept. 30, 1996 (444K PDF)
|
 |
|
Dr. E. Donnal Thomas's letter to state investigator, his only public statement ever on Protocol 126, Sept. 19, 1996 (608K PDF)
|
|

